Exploring the Impact of Chiropractic Intervention in the Quality of Life in Postural Orthostatic Tachycardia Syndrome: A Case Report Brandon T Brock, DC
Presented to the Craniocervical Specialist diplomate program 2025
Abstract
Postural Orthostatic Tachycardia Syndrome (POTS) is a disorder that may stem from the central nervous system according to recent literature and is a diagnosis of exclusion, so other disorders must be ruled out first. POTS is defined by an increase in heart rate ≥ 30 bpm within 10 minutes of a change from the supine position to an upright position (or during a head-up tilt test to at least 60°), in the absence of orthostatic hypotension. I present a case of a 34 year old female with over a decade of dizziness upon standing or with movement/exercise, migraine headaches, depression, neck and back pain, presyncope, brain fog, lightheadedness, chest pain, shortness of breath and anxiety. Previous examinations ruled out other conditions and has a diagnosed condition of POTS from other medical providers. Cone beam computed tomography showed an articular misalignment of the first cervical vertebra. Over three months, the patient underwent chiropractic care to determine if we could improve her quality of life. The SF-36 was utilized to determine if we could improve her quality of life and she showed an improved score in most categories. This case highlights the importance of considering craniocervical dysfunction with associated conditions like POTS. While she showed an improved quality of life from the conservative management of chiropractic care, additional research is needed on this topic.
What is POTS?
Originally identified in 1982 and may have been described as early as 1871 as dacosta syndrome or “an irritable heart” (DaCosta 1871) and characterized further in 1993 (Schondorf and Low, 1993)Most studies focus their diagnostic criteria focus on the heart rate and fail to capture the variety of other symptoms that are a part of this syndrome. (Raj et al. 2019)POTS has been defined by major international neurologic, autonomic, cardiac and pediatric societies (Freeman et al., 2011; Raj et al., 2020; Sheldon et al., 2015) as requiring: 1. A sustained HR increment of not less than 30 beats/minute within 10 min of standing or head-up tilt. For individuals who are 12 to 19 years old, the required HR increment is at least 40 beats/minute2. An absence of orthostatic hypotension (i.e. no sustained systolic blood pressure [BP] drop of 20 mmHg or more)3. Frequent symptoms of orthostatic intolerance during standing, with rapid improvement upon return to a supine position. Symptoms may include lightheadedness, palpitations, tremulousness, generalized weakness, blurred vision, and fatigue4. Duration of symptoms for at least 3 months5. Absence of other conditions explaining sinus tachycardia such as anorexia nervosa, primary anxiety disorders, hyperventilation, anemia, fever, pain, infection, dehydration, hyperthyroidism, pheochromocytoma, use of cardioactive drugs (e.g. sympathomimetics, anticholinergics) or severe deconditioning caused by prolonged bed rest. (Vernino et al. 2019)The etiology of POTS is largely unknown and debated. 90% female predominance possibly linked to a smaller upright stroke volume compared to men. Affecting .1%-1% or 0.5-3 million individuals. 25% are unable to work as a result of their disability (Grubb 2008).The natural history of POTS is not clear, but it does not appear to increase the risk of mortality (Sheldon et al. 2015)
Common Symptoms
Orthostatic symptoms, such as rapid palpitations, lightheadedness, chest discomfort, and dyspnea, and non cardiac symptoms, such as mental clouding, headache, nausea, tremulousness, generalized weakness, and blurred or tunnel vision, are evident in POTS patients during upright posture (Raj 2019)Non-Orthostatic or also described as autonomic problems in POTS might include gastrointestinal complaints, such as abdominal pain, nausea and irritable bowel syndrome, as well as bladder symptoms and abnormal sudomotor regulation, and more generalized complaints in patients with POTS include hypermobile joints, exercise intolerance, migraine headaches, sleep disturbances, and fatigue (Raj 2019)(Theiben et al. 2007)
Common comorbidities: Chronic fatigue syndrome, ehlers danlos syndrome, vasovagal syncope, inappropriate sinus tachycardia (Garland et al 2015). The orthostatic tachycardia of POTS is exaggerated in the morning compared to the evening. To optimize diagnostic sensitivity, it is recommended that postural testing be performed in the morning (Brewster et al. 2012)The observation of orthostatic tachycardia is not sufficient for a diagnosis of POTS. The HR abnormality must be accompanied by chronic (>3-6 months) orthostatic symptoms that occur in the absence of prolonged bed rest or deconditioning, use of medications that impair autonomic regulation (vasodilators, diuretics, antidepressants, or anxiolytic agents), or other chronic debilitating disorders that might cause tachycardia (such as dehydration, anemia, or hyperthyroidism). A single negative tilt table test does not necessarily exclude POTS either (Raj 2013) (Huh et al. 2013) and depending on the time length used for test it can increase the chances for false positives. Standing tests activate a skeletal muscle pump which decreases likelihood on a positive test (Plash et al. 2014)The clinical evaluation of patients with suspected POTS is not standardized, nor are treatment approaches. There is a lack of familiarity with POTS in the medical community, and the epidemiology of the disorder and natural history are not known(Raj and Robertson, 2018)There is a critical need for an efficient, cost-effective, multidisciplinary approach, with emphasis on the “medical home” (Jackson et al., 2013). Importantly, such an approach has the potential to improve efficiency of clinical care, patient experience, and lower medical costs (Williams et al., 2012)
POTS is a variant of autonomic cardiovascular disorder and a common type of dysautonomia. (Olshansky et al 2020)First is the role of genetic factors in defining the risk and determining the manifestation of POTS. A monogenetic cause is unlikely, but a familial tendency exists. The associations of POTS with migraine headache, the hypermobile form of Ehlers-Danlos syndrome (hEDS) and the hypermobility spectrum disorder, allergic disorders like mast cell activation syndrome (MCAS), and autoimmune disorders have been clearly described. Prior studies have documented small fiber neuropathy in approximately half of POTS patients. There is some evidence that small fiber neuropathy in POTS may correlate to reduced cardiac innervation (Raj, Satish R. et al.2019)Most of the postganglionic sympathetic innervation to the heart descends from the middle cervical ganglion at C5. Somatic dysfunction at C6 may influence both the pre- and postganglionic fibers and control of the heart rate. Attenuation and even blocking of afferent baroreceptor activity by somatic afferents has been described. In addition, modulation of posturally induced systemic vasoconstriction by upper cervical somatic afferent activity interacting with vestibular activity has been reported. Cranial and upper cervical somatic dysfunction may adversely affect both baroreceptor afferent function and posturally induced vasoconstriction. (Goodkin and Bellew 2014)(Budgell 2011)In the hyperadrenergic state and distal neuropathy are involved in the pathophysiology of POTS, “certain features suggest brainstem dysregulation” (Low et al. 1998)Some patients may have craniocervical instability, which manifests with cerebrospinal fluid flow obstruction and compression of the medulla or pons where central autonomic nervous system networks are located. (Blitshteyn 2021)At the level of dorsal medulla, higher total choline and myo inositol were found on magnetic resonance (MR) spectroscopy in adolescents with POTS, syncope and orthostatic hypotension suggestive of neuroinflammation which is the underlying cause of autonomic dysregulation as compared to healthy controls (Wagoner et al. 2019)
No clear pathologic substrate and multiple interacting pathophysiologic mechanisms. Thus, it resembles functional visceral pain/dysmotility disorders, fibromyalgia, chronic headache, and chronic fatigue syndrome. Symptoms frequently develop after a triggering factor such as a viral illness or surgical procedure and persist despite resolution of the underlying condition. Chronicity depends on common interacting mechanisms that likely reflect plastic changes in central nervous system areas involved in processing of interoceptive (visceral and somatic nociceptive) information, interoceptive awareness, behavioral arousal, and stress responses.(Benarroch 2012)
3 main types are consistent in the literature and some identify up to 5.Symptoms may vary patient to patient as well as subtype to subtype and will have some form of overlap in subtypes/symptoms (Garland et al 2015)The clinical relevance of these subtypes is unknown due to lack of uniform phenotyping, and the fact that patients may present with multiple overlapping features. It remains uncertain if these represent different underlying diseases or if they are different manifestations of a common underlying disease (Vernino et al., 2021). 1 in 2 patient with POTS will have neuropathic features 1 in 3 patients will have hyperadrenergic features 1 in 7 will have serological evidence of an autoimmune pathogenesis(Thieben et al. 2007)
Hyperadrenergic POTS is characterized by elevated levels of plasma norepinephrine levels (neurotransmitter of thesympathetic NS) >600 pg/mL and >10 mmHg rise in systolic blood pressure while standing for 10 minutes (Low et al. 2009).In hyperadrenergic POTS, the sympathetic nervous system is overactivated.The most prominent symptoms associated with hyperadrenergic POTS include: Increased blood pressure, Tachycardia thatmay be more extreme than other subtypes, Heart palpitations, Anxiety, tremor, cold, sweaty extremities while upright (Lowet al. 2009), Migraine headaches, Increased urinary output after being upright, Nausea/vomitingThe most common comorbidity with hyperadrenergic POTS is mast cell activation syndrome (Wang et al. 2021). Identifyingand treating mast cell activation syndrome, if present, can help to decrease these symptoms. There are a number of otherpotential causes of hyperadrenergic POTS, which are rare. These include a single point mutation in the norepinephrinereuptake receptor (Lambert et al. 2008), baroreflex failure, and a tumor on the adrenal glands called pheochromocytoma(Benarroch 2012).Treatments for hyperadrenergic POTS focus on rebalancing the autonomic nervous system, typically by decreasing theactivity of the sympathetic nervous system. These medications include: Clonidine - central nervous system sympatholyticmedication that decreases the central sympathetic tone, Beta Blockers - block B-adrenergic receptors bound bynorepinephrine and prevent sympathetic activity peripherally (Fedorowski 2018).
Neuropathic POTS is characterized by a decrease in sympathetic innervation, particularly in the legs (Mar and Raj 2020), and is often associated with small fiber neuropathy. Normally, these small fiber nerves regulate constriction of the blood vessels in the limbs and abdomen. Because these nerves are damaged in neuropathic POTS, however, activation of the sympathetic nervous system leads to less release of norepinephrine from these small fibers. Because norepinephrine is a vasoconstrictor, neuropathic POTS is often associated with excessive blood pooling in the hands and feet. This decreases blood circulation and causes less blood to return to the heart (low venous return) which causes the heart rate to increase in order to compensate. Neuropathic POTS often occurs following infection, surgery or trauma and may be an autoimmune form of POTS, as autoantibodies to the ganglionic acetylcholine receptor have been associated with this POTS subtype (Li et al. 2014). A thermoregulatory sweat test or a Quantitative Sudomotor Axon Reflex Test QSART), quantitative sensory testing, or a skin biopsy can also be used to determine whether an autonomic neuropathy exists (Miglis, Muppidi 2017)The most prominent symptoms of neuropathic POTS include:Loss of sweating in extremitiesBlood poolingCyanosis in the feet (turning a bluish color when standing/warm)Treatments for neuropathic POTS tend to focus on decreasing blood pooling and moving blood back up toward the heart and brain. Theseinclude:Graded compression garments, including abdominal compression of 40 mmHg (Mar and Raj 2020) and leg compression of20-30 mmHg.Midodrine – an alpha 1 adrenergic agonist (binds NE receptors) that causes constriction of the peripheral blood vessels andpromotes return of blood to the heart and brain (Mar and Raj 2020).Pyridostigmine – an acetylcholinesterase inhibitor that increases the acetylcholine levels in the synapse (increasingparasympathetic activity) of autonomic ganglia (Mar and Raj 2020) which helps to decrease heart rate.
Hypovolemic POTS is characterized by abnormally low blood levels, including both red blood cells and plasma (Raj et al. 2005). Whilenot often measured, the plasma deficit in POTS is approximately 13% and may be related to low levels of circulating renin andaldosterone which are fundamental for the regulation of blood volume (Raj et al. 2005)(Garland et al. 2015). As a result, people with thistype of POTS have their kidneys excreting too much sodium and water, which contributes to this low blood level. Low blood volumeleads to less blood return to the heart, which causes an increased heart rate and force of contraction in an attempt to keep bloodcirculation at normal levels.What symptoms are typically associated with hypovolemic POTS?WeaknessDecreased tolerance for exerciseWhat are the typical non-pharmacological treatments?Increased salt (6-10 g) and fluids (2-3 L) to increase blood volume (Fu and Levine 2018)Compression garments with at least 20-30 mm Hg pressure, including leg stockings and a pair of biker-style shorts (Fu andLevine 2018)Raise head of bed 4-6 inches. Mild orthostatic stress shifts fluid to the lower body which activates therenin-angiotensin-aldosterone system, leading to fluid retention at the kidneys and blood volume expansion (Fu and Levine2018).What are the typical medications to increase blood volume? Fludrocortisone: enhances sodium and water retention to increase bloodvolume, Desmopressin: synthetic version of antidiuretic hormone that increases water reabsorption without increasing sodiumreabsorption to increase blood plasma levels, Erythropoietin: stimulates red blood cell production and increases overall blood volume, IVfluids can be helpful.
Current Therapies or treatment options
Currently, no medications are approved by the U.S. Food and Drug Administration (FDA) specifically for POTS. Pharmacologic therapies are primarily used to manage specific symptoms. (Garland et al 2015)The treatment of POTS should start with non-pharmacological treatments, the Vanderbilt Approach (Garland 2015) Patients are encouraged to increase their blood volume by drinking 2–3 L/day of water and ingesting >200 mEq sodium/day (Heart Rhythm Society (HRS) 2015 Statement Class IIb recommendation)Intravenous saline (1–2L) can quickly expand blood volume and lessen tachycardia in POTS patients (Jacob et al. 1997) but for an acute clinical decompensation in POTS only, Abdominal compression with elastic waist high support hose and abdominal binders can help decrease splanchnic-mesenteric venous pooling during standing (Sheldon et al. 2015)The three-month Dallas Program, which involves aerobic exercise coupled with resistance training, improves orthostatic and exercise tachycardia, symptoms, and quality of life and improvement is related to an increase in the size of the heart and increased blood volume. The improved stroke volume decreases tachycardia. (Sheldon et al. 2015)The challenge with an exercise program in POTS is the marked exercise intolerance and debilitating post-exercise fatigue. The Dallas Program focuses on starting with seated or supine exercises (e.g., rowing machine, recumbent cycle, or swimming) three times per week. Patients must be informed that it can take 5–6 weeks before they notice any improvement, and in the interim, the patients may transiently feel worse. A structured and progressive exercise program is a HRS Class IIa recommendation (Sheldon et al. 2015)
A systematic review and meta-analysis showed out of all the studies included in the criteria that 5 studies were of high quality. Of the included high quality studies there were oral water/salt intake, exercise, midodrine, cardioselective b-receptor blockers, non cardioselective b-receptor blockers; Other included studies within the same categories did not show the same high quality results. (Wells et al. 2018)The treatment of POTS is difficult; there are no therapies that are uniformly successful, and combinations of approaches are often needed. Few treatments have been tested with the usual rigor of randomized clinical trials, and there is no consensus as to whether specific treatments should be targeted to subsets of POTS or whether a uniform approach should be used. (Sheldon et al. 2015)
Cases reports in the past
Chiropractic shows promising results from current studies on autonomic function (Kiani et al. 2020)A 27-year-old woman with a six-year history of dizziness, neck and cervicothoracic pain, and occipital-temporal-orbital headaches. The patient also experienced occasional severe, incapacitating episodes of dizziness with vomiting which has been occurring for over 6 years. Previous evaluations, including advanced imaging, had helped rule out central, otolithic, and psychogenic causes of dizziness.eight months, the patient underwent a regimen of gentle manual therapies and cervical stability exercises. The patient’s dizziness substantially improved, as measured by the Dizziness Handicap Inventory (DHI), with scores reducing from 50 (moderate handicap) to 10 (less than mild handicap). (Trager et al. 2024)A 50-year-old female presented with lightheadedness, palpitation, and neck pain for 3 years. Following 3 months of regular treatment, the patient reported a full resolution of neck pain, dizziness, and POTS. At 12-month follow-up, all radiographic metrics showed improvement, including improvement of cervical alignment and thoracic curvature. The patient’s neck pain decreased from 5/10 to 0/10 on the numeric pain rating scale as a result of treatment. The DHI score decreased from 46 to 0 in this case. (Chu and Lin 2022)
A 26-year-old woman presented to the cardiology department of the hospital with a 6-year history of fatigue, presyncope, severe heat intolerance, cognitive dysfunction, diffuse joint pain, and insomnia. The patient denied any precipitating event but related injuring her jaw when she fell during an obstacle course. At 18-month follow-up, the patient reported tolerating a hot shower without symptoms and standing for hours as long as she was able to shift her weight. Her fatigue, energy, and concentration were much improved. She no longer experienced presyncope and had minimal joint pain. Her main symptom was getting overheated. In the supine position, the patient had a baseline blood pressure of 120/74 mm Hg and a heart rate of 80 beats per minute. During a repeated standing test, she had no orthostatic symptoms during 10 minutes of standing, and blood pressure and heart rate were stable. The patient's retrospective self-rated Wellness score was 40 out of 100 before presentation, with 0 representing death and 100 representing as good as a person could feel. At 18-month follow-up, the patient rated her well-being as 90. (Goodkin and Bellew 2014)The patient is a 31-year-old Caucasian female previously diagnosed with postural orthostatic tachycardia syndrome by her cardiologist when she was 18. Her symptoms were so bad that she was placed on long term bed rest. She also suffered from chronic neck pain. The patient was assessed during a chiropractic examination which found multiple vertebral subluxations in the cervical, thoracic, lumbar, and pelvic regions of the spine. Thermal scanning revealed dysautonomia. Diversified Technique was used to analyze and adjust the patient over the course of 26 weeks where she recorded 45 visits to correct the vertebral misalignments in the spine. The patient reported a decrease in the occurrence and severity of her postural orthostatic tachycardia syndrome along with decrease in her musculoskeletal complaint of neck pain. Thermographic scans revealed a decrease in dysautonomia. (Wilson 2022)
A 36-year-old female sought chiropractic care for POTS induced nausea/dizziness/palpitations, constant bilateral occipital headaches, neck and sacroiliac/sciatic pain, exercise intolerance and bloating. Patient history, physical examination, and active 10-minute stand test established that she met the criteria for POTS.Craniocervical and spinal misalignments, believed possibly leading to cerebral vasculature and connective tissue deformation, were corrected according to the Sacro-Occipital Technique and Atlas Orthogonal upper cervical evaluation protocols. Specific POTS related therapeutic exercises, diet modifications, as well as ocular fixation exercises were also implemented. Orthostatic vital sign testing was regularly recorded to assess POTS patient’s response to care. Following combined treatment and adjunct therapy, the patient no longer met the criteria for POTS, did not exhibit pain or orthostatic intolerance symptoms to postural change except for mild brief bouts of dizziness. (Vitez et al. 2022)Two patients, a sixty two year old male diagnosed with Paroxysmal Supraventricular Tachycardia (PSVT) and a twenty two year old female diagnosed with Postural Orthostatic Tachycardia Syndrome (POTS) were evaluated. Both patient's’ chief complaint was low back pain. POTS patient also had a history of migraine. Both had a history of significant head trauma.Five months after initial adjustment, PVST patient presented with absence of pattern, change in HRV and resolution of lumbago. POTS patient presented nine weeks after first adjustment migraine free. Her POTS was unchanged. Her HRV analysis was unchanged.Sometimes higher heart rate variability is not better heart rate variability. In these two cases there appears to be a relationship between the improvements in bilateral skin temperature pattern analysis and Prill modified tests with over all health following an upper cervical chiropractic technique. It is feasible that upper cervical chiropractic care can have a positive effect on the autonomic nerve system. Further study is recommended to determine the role of HRV testing in subluxation management. (Burcon 2016)
All the studies show promising results with various methods of chiropractic techniques and some included other therapies such as cervical strengthening exercises and most showing self reported improvements to symptoms associated with POTS from pain, dizziness, migraine headaches or surveys showing decreases and others performing standing tests showing the patient did not meet the criteria for POTS at that time. One of the studies showed improvement to symptoms that are comorbid with POTS but did not have improvements in the condition of POTS.These are all the published studies I was able to find that included spinal manipulation from chiropractors to osteopathic medicine doctors from pudmed, parker, shermans research journals and the journal for vertebral subluxation research and scihub.
Case report at the Craniocervical Care Center
34 year old female that reports no precipitating factors immediately before symptoms occurred. She reports about 5 years prior that the only significant accident was hitting her head into a brick wall in high school while playing sports. She reports that the symptoms became much worse over the past year before our care started after contracting covid with influenza type A. Symptoms include: dizziness upon standing or with movement/exercise, migraine headaches, depression, neck and back pain, presyncope, brain fog, lightheadedness, chest pain, shortness of breath, anxiety and she has had these symptoms for over 10 years.She also reports she has seen multiple doctors on and off for a decade without success - Primary medical doctor, Physical Therapist, Chiropractor, Neurologist, ENT (ear, nose throat doctor), Cardiologist, Endocrinologist, Functional medicine doctor and holistic health coach.
As many more practitioners are identifying and diagnosing POTS, there have been many patients with POTS seeking upper cervical care. In my office, I have seen many cases of dysautonomia, POTS-like symptoms, patients with orthostatic tachycardia and orthostatic hypotension and POTS POTS-like patients have not received a formal diagnosis and report many symptoms that may appear as POTSPatients in my office that I have seen with a POTS diagnosis are about ten.POTS is a common dysautonomia and even though its common, there is very little understanding in the pathophysiology and little to no standard treatment protocols. To me, it is fascinating that all the current research is identifying the brainstem and the more research we are able to do with our image guided craniocervical correction This case was selected because the patient had a confirmed diagnosis of POTS without secondary factors, as well as they were the only patient to fill out a pre and post survey within a 3 month period. All others were excluded since we are focusing on the short term changes in the quality of life.
Our active care consisted of eleven visits over three months. The patient was given chiropractic adjustments only. If a craniocervical adjustment was indicated then that was the only intervention received on a given day and if not indicated then full spine adjustments were performed. Over the course of the three months the patient received a total of 6 craniocervical adjustments using the blair upper cervical technique. Five of the visits full spine adjustments varied from the thoracic, lumbar and sacrum with an activator. Our adjustments were performed in accordance with the Blair and Activator protocols separately. For the Blair protocol, the patient was placed in a neutral side lying posture on a lloyd toggle table, correction vectors were obtained from the imaging and utilized in the hand and stance position of the doctor. The headpiece would drop as the correction occurred.The doctor delivering the blair upper cervical adjustment is certified proficient in the technique.The Sf-36 was chosen at the time since this survey is one of the most widely used general health status measures and has been many times validated and is a great general gauge for overall health as well as being able to identify which areas in the patient's life are improving.(Brazier 1992)
Cervical distraction - relief of pain, Foraminal compression - straight down compression WNL, R side FC caused radiating pain from the R side C1 to the R shoulder and L FC caused localized pain in the mid cervical, Jackson's compression - WNL, Valsalva - pressure in neck and R shoulder blade area, Muscle strength C5-T1 and L2-S1 innervations 5/5, sensory nerve testing C5-T1 and L3-S2 WNL, CN XII - Tongue Deviation -Left, Fasciculation - Present, Scalloping - Absent, CN IX, X - Symmetrical elevation of the soft palate when saying 'Ahh' and reports not aspirating food or water often as well as normal cough and no hoarse voice or feeling of something stuck in throat. CN VII - Normal, CN V - WNL, Pupil Size - Normal Findings, Pupillary Reflex Left and right - WNL, Capillary Refill Time Left and right - Normal, Fukuda Step Test - WNL, VOR - R side horizontal beating nystagmus but maintained focus and L side multiple attempts patient could not focus on target and moderate to severe dizziness with both sides.Re-Exam 2 months later showed all the above tests were normal except for Foraminal compression R FC caused pain along the L side of the neck
CBCT 3D to determine if there is an articular misalignment in the CCJBlair Analysis - L protracto view PIR, R protracto view ASR. Listing - ASRPIR 43/31
DIAGNOSTIC IMAGING REPORT CONE BEAM CT (CBCT) EXAMINATION – UPPER CERVICAL SPINE INDICATIONS: Images received for secondary radiographic consultation. No history provided. TECHNIQUE: 0.3-mm axial slices from the skull base to C5/6 were obtained. Coronal and sagittal digital reformation was rendered. COMPARISON: None. FINDINGS: No evidence of acute fracture or dislocation. The cervical lordosis measures 8° (Normal 35°-45°) utilizing the superior endplate of C3 and the inferior endplate of C6. Overall osseous density is maintained. Bilateral stylohyoid ligament ossification without approximation to C1. No evidence of prevertebral soft tissue swelling. The temporomandibular joints are maintained. Craniocervical Junction: The visualized skull bases are normal. The mastoid air cells are well aerated. The atlantodental interval and paraodontoid spaces are within normal limits. The clivoaxial angle measures approximately 149° (Normal: 145-160°). Chamberlain line is within normal limits. Normal atlantodental interval. Lower cervical spine: C2-3: Vertebral body and intervertebral disc heights are relatively maintained. No evidence of osseous neuroforaminal or spinal canal stenosis. C3-4: Vertebral body and intervertebral disc heights are relatively maintained. No evidence of osseous neuroforaminal or spinal canal stenosis. C4-5: Vertebral body and intervertebral disc heights are relatively maintained. No evidence of osseous neuroforaminal or spinal canal stenosis. C5-6: Vertebral body heights are relatively maintained. Mild loss of disc height with endplate spurring. Large posterior osteophyte, mildly narrowing the spinal canal. Mild uncovertebral spurring. No evidence of osseous neuroforaminal stenosis. Paranasal sinuses: Mild mucosal thickening in the bilateral maxillary sinuses. The nasal septum is near midline without spur. IMPRESSIONS: 1. No acute fracture or dislocation. 2. Mild discogenic spondylosis at C5/6. 3. Mild uncovertebral arthrosis at C5/6. 4. Mild spinal canal stenosis at C5/6. 5. Mild mucosal thickening in the bilateral maxillary sinuses. 6. Bilateral stylohyoid ligament ossification without approximation to C1. 7. Flattening of the cervical lordosis
Scoring the RAND 36-Item Health Survey is a two-step process. First, precoded numeric values are recoded per the scoring key given in Table 1. Note that all items are scored so that a high score defines a more favorable health state. In addition, each item is scored on a 0 to 100 range so that the lowest and highest possible scores are 0 and 100, respectively. Scores represent the percentage of total possible score achieved. In step 2, items in the same scale are averaged together to create the 8 scale scores. Table 2 lists the items averaged together to create each scale. Items that are left blank (missing data) are not taken into account when calculating the scale scores. Hence, scale scores represent the average for all items in the scale that the respondent answered.
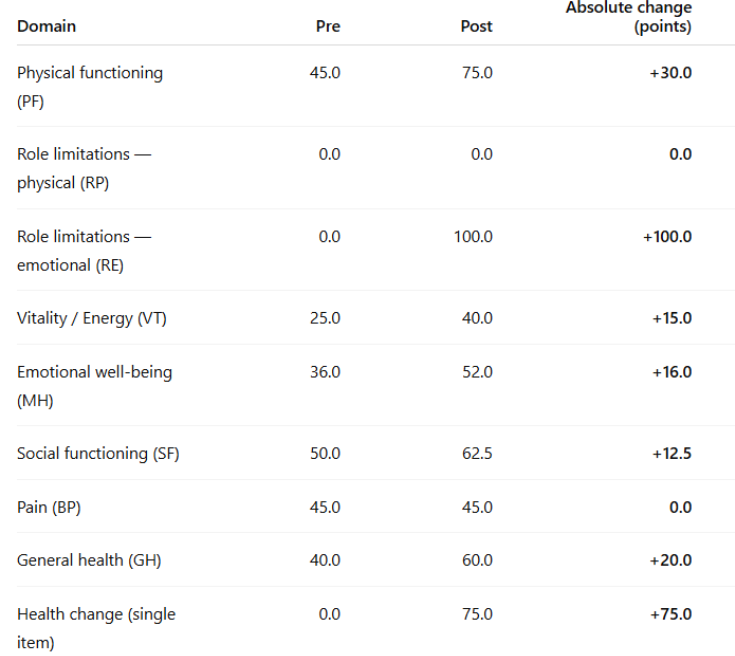
The physical functioning domain reflects a participant’s capacity to perform basic physical activities such as walking, climbing stairs, lifting, and self-care. In this study, scores increased from 45 at baseline to 75 post-intervention, representing a substantial 30-point improvement. This shift indicates that participants experienced meaningful gains in mobility and independence, moving from severe limitations toward a level of functioning closer to population norms. Improvements of this magnitude suggest enhanced ability to carry out daily activities and greater physical autonomy, which are often considered central indicators of recovery and rehabilitation success. These findings align with prior research showing that interventions targeting strength, endurance, or neuromusculoskeletal function can produce significant improvements in this domain, even when other areas such as pain or fatigue remain less responsive.
The role limitations due to physical health domain of the assesses the extent to which physical health interferes with work or other daily role responsibilities, such as accomplishing tasks, meeting time demands, or performing duties as expected. In this study, scores remained unchanged at 0 both pre- and post-intervention, indicating that despite improvements in physical functioning, participants continued to experience substantial restrictions in fulfilling occupational or role-related activities. This lack of change highlights the distinction between physical capacity and role participation: while individuals may regain the ability to perform specific tasks, persistent symptoms, fatigue, or external constraints may still prevent them from meeting role expectations.
The role limitations due to emotional problems domain measures the extent to which emotional difficulties, such as anxiety or depression, interfere with work or other daily responsibilities. In this study, scores improved markedly from 0 at baseline to 100 post-intervention, indicating a complete resolution of emotional barriers to role participation. This dramatic change suggests that participants no longer perceived emotional distress as limiting their ability to fulfill occupational or social responsibilities. Improvements of this magnitude are uncommon and highlight the potential impact of the intervention on psychological resilience and coping.
The energy or vitality domain assesses perceived energy levels and fatigue, providing insight into participants’ overall sense of vigor and stamina. In this study, scores increased moderately, from 25 pre-intervention to 40 post-intervention, indicating 15-point improvement. While this change suggests a moderate reduction in fatigue, it represents a meaningful shift in daily energy or endurance. The persistence of low vitality despite significant gains in physical functioning and emotional role participation highlights the complex and multifactorial nature of fatigue, which is often influenced by pain, sleep quality, mood, and physiological regulation.
The emotional well-being domain evaluates general mental health, including symptoms of anxiety, depression, and positive affect. In this study, scores improved from 36 at baseline to 52 post-intervention, representing a 16-point gain. This meaningful increase suggests that participants experienced reductions in emotional distress and improvements in psychological well-being. While not as dramatic as the complete resolution of role limitations due to emotional problems, the improvement in overall emotional health points to a broader enhancement in mood stability and coping capacity, which may contribute to sustained quality-of-life benefits over time and has some clinical significance.
The emotional well-being domain evaluates general mental health, including symptoms of anxiety, depression, and positive affect. In this study, scores improved from 36 at baseline to 52 post-intervention, representing a 16-point gain. This meaningful increase suggests that participants experienced reductions in emotional distress and improvements in psychological well-being. While not as dramatic as the complete resolution of role limitations due to emotional problems, the improvement in overall emotional health points to a broader enhancement in mood stability and coping capacity, which may contribute to sustained quality-of-life benefits over time and has some clinical significance.
The social functioning domain reflects the degree to which physical and emotional health interfere with normal social activities, such as visiting friends, engaging in community events, or maintaining relationships. Scores rose from 50 pre-intervention to 62.5 post-intervention, a 12.5-point improvement. This suggests that participants were better able to participate in social interactions and felt less restricted by their health conditions in social contexts. Improvements in this domain are particularly important, as social connectedness is strongly associated with emotional resilience and long-term health outcomes.
The pain domain measures both the intensity of bodily pain and the extent to which pain interferes with daily activities. In this study, scores remained unchanged at 45.0 both pre- and post-intervention, indicating no measurable reduction in pain severity or its impact on functioning. This finding contrasts with the substantial gains observed in physical functioning, suggesting that participants were able to increase activity levels and independence despite persistent pain. The stability of pain scores underscores the importance of distinguishing between functional recovery and symptom relief, and highlights the need for targeted pain management strategies or co-management in future interventions.
The general health domain assesses participants’ overall perception of their health, including current status and expectations for future health. Scores improved from 40 at baseline to 60 post-intervention, representing an 20-point increase. This improvement reflects a more optimistic outlook on health and greater confidence in overall well-being, suggesting that participants not only experienced functional gains but also perceived these gains as meaningful to their quality of life. Such improvements in self-rated health are clinically significant, as perceived health status is a strong predictor of long-term outcomes, including morbidity and mortality.
The health change item provides a global measure of perceived improvement or decline in health over the past year. Scores increased from 0.0 to 75.0 post-intervention, reflecting a large positive shift in participants’ retrospective evaluation of their health trajectory. This result suggests that participants overwhelmingly perceived themselves as healthier compared to the previous year, providing strong subjective evidence of intervention effectiveness. Although this item is distinct from the other domains, its substantial increase reinforces the positive trends observed across physical and psychosocial health measures.
These findings highlight both the successes and limitations of the intervention. Substantial improvements in physical functioning, emotional role participation, perceived health, and social engagement demonstrate that the program produced clinically significant benefits. At the same time, the persistence of pain and physical role limitations underscores the need for targeted strategies to address these barriers. Future interventions should consider integrating multidisciplinary approaches in order to translate functional gains into sustained improvements across all domains of quality of life.The developers of the SF-36 have suggested that using the general health dimension a five-point difference (on the 0–100 scale) is the smallest score change achievable by an individual and considered as 'clinically and socially relevant'. (Ware et al. 1993)There is clinical significance in this case study
At 6 months, the patient reports that her POTS symptoms seem to coincide with her upper neck needing a Blair upper cervical correction. This seems to occur once a month or so and there will be other symptoms such as neck and/or back pain that are more consistent but an upper neck correction will not be indicated.This may lead to POTS coming from a brainstem type disorder as indicated earlier in the research.Further studies are necessary to determine if POTS may truly be a brainstem disorder and if upper cervical chiropractic should be a initial treatment in conjunction with other proposed therapies such as PT, fluids and electrolytes.
Currently, we are waiting on IRB approval for a prospective case series for patients with a confirmed diagnosis of POTS to receive craniocervical corrections only.
Discussion
Several limitations should be considered when interpreting these findings. First, the sample size and variability were not reported here, which limits the ability to calculate statistical significance or effect sizes for the observed changes.The study design did not include a control group, making it difficult to determine whether the improvements observed were due to the intervention itself or to natural recovery, placebo effects, or other external influences.Limited research on subtypes of POTS and many patients show signs of multiple subtypes. There was not a subtype identified on this study and all pots patients may not respond the same.Study was not limited to image guided craniocervical adjustments only since we included full spine activator. The patient did report she was not taking any prescribed medication but was taking a variety of supplements which changed monthly from her functional medicine doctor and holistic health coach.
Study did not include multiple standing or tilt table tests. We did not track progress as far her orthostatic intolerance and only focus on quality of life so there Study did not include other forms of surveys or ways to track progress that would be helpful with POTS specific symptoms like the Malmo POTS scoreA number of the published randomized interventional studies and this study consist of acute interventions aimed at demonstrating short term improvement of tachycardia and orthostatic tolerance. Longitudinal studies are needed in our field of upper cervical chiropractic to determine the long term effectiveness with larger case series. More research is needed.
Cited Studies / Sources
Rachel Wells, MBBS; Adrian D. Elliott, PhD; Rajiv Mahajan, MD, PhD; Amanda Page, PhD; Valeria Iodice, MD, PhD; Prashanthan Sanders, MBBS, PhD; and Dennis H. Lau, MBBS, PhD, Efficacy of Therapies for Postural Tachycardia Syndrome: A Systematic Review and Meta-analysis, 2018 Mayo Foundation for Medical Education and Research n Mayo Clin Proc. 2018;nn(n):1-11
Grubb BP. Postural tachycardia syndrome. Circulation. 2008;117: 2814–7.Garland EM, Celedonio JE, Raj SR. Postural Tachycardia Syndrome: Beyond Orthostatic Intolerance. Curr Neurol Neurosci Rep. 2015 Sep;15(9):60. doi: 10.1007/s11910-015-0583-8. PMID: 26198889; PMCID: PMC4664448.
Wells R, Elliott AD, Mahajan R, Page A, Iodice V, Sanders P, Lau DH. Efficacy of Therapies for Postural Tachycardia Syndrome: A Systematic Review and Meta-analysis. Mayo Clin Proc. 2018 Aug;93(8):1043-1053. doi: 10.1016/j.mayocp.2018.01.025. Epub 2018 Jun 21. PMID: 29937049.
Olshansky B, Cannom D, Fedorowski A, Stewart J, Gibbons C, Sutton R, Shen WK, Muldowney J, Chung TH, Feigofsky S, Nayak H, Calkins H, Benditt DG. Postural Orthostatic Tachycardia Syndrome (POTS): A critical assessment. Prog Cardiovasc Dis. 2020 May-Jun;63(3):263-270. doi: 10.1016/j.pcad.2020.03.010. Epub 2020 Mar 25. PMID: 32222376; PMCID: PMC9012474.
Benarroch EE. Postural tachycardia syndrome: a heterogeneous and multifactorial disorder. Mayo Clin Proc. 2012 Dec;87(12):1214-25. doi: 10.1016/j.mayocp.2012.08.013. Epub 2012 Nov 1. PMID: 23122672; PMCID: PMC3547546.Postural orthostatic tachycardia syndrome (POTS): Priorities for POTS care and research from a 2019 National Institutes of Health Expert Consensus Meeting – Part 2, Raj, Satish R. et al., Autonomic Neuroscience: Basic and Clinical, Volume 235, 102836
Sheldon RS, Grubb BP, Olshansky B, Shen WK, Calkins H, Brignole M, et al. 2015 heart rhythm society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm. 2015;12:e41–63.
S. Vernino, K.M. Bourne, L.E. Stiles, et al., Postural orthostatic tachycardia syndrome (POTS): State of the science and clinical care from a 2019 National Institute of Health Expert Consensus Meeting, Autonomic Neuroscience: Basic and Clinical (2018), https://doi.org/10.1016/j.autneu.2021.102828
Fu Q, Levine BD. Exercise and non-pharmacological treatment of POTS. Auton Neurosci. 2018 Dec;215:20-27. doi: 10.1016/j.autneu.2018.07.001. Epub 2018 Jul 4. PMID: 30001836; PMCID: PMC6289756.Chu ECP, Lin AFC. Relief of postural orthostatic tachycardia syndrome with chiropractic rehabilitation. J Family Med Prim Care. 2022 Jul;11(7):4006-4009. doi: 10.4103/jfmpc.jfmpc_2108_21. Epub 2022 Jul 22. PMID: 36387679; PMCID: PMC9648254.
Trager RJ, Schuster A, Tao C, Zamary G. Conservative Management of Cervicogenic Dizziness Associated With Upper Cervical Instability and Postural Orthostatic Tachycardia Syndrome: A Case Report. Cureus. 2024 Oct 31;16(10):e72765. doi: 10.7759/cureus.72765. PMID: 39618563; PMCID: PMC11608111.
Jacob G, Shannon JR, Black B, Biaggioni I, Mosqueda-Garcia R, Robertson RM, Robertson D. Effects of volume loading and pressor agents in idiopathic orthostatic tachycardia. Circulation. 1997 Jul 15;96(2):575-80. doi: 10.1161/01.cir.96.2.575. PMID: 9244228.
Stewart JM, Medow MS, Glover JL, Montgomery LD. Persistent splanchnic hyperemia during upright tilt in postural tachycardia syndrome. Am J Physiol Heart Circ Physiol. 2006;290:H665–73
Fu Q, Vangundy TB, Galbreath MM, Shibata S, Jain M, Hastings JL, et al. Cardiac origins of the postural orthostatic tachycardia syndrome. J Am Coll Cardiol. 2010;55:2858–68.Kiani AK, Maltese PE, Dautaj A, et al. Neurobiological basis of chiropractic manipulative treatment of the spine in the care of major depression. Acta Biomed. 2020;91(13-S):e2020006. Published 2020 Nov 9. doi:10.23750/abm.v91i13-S.10536
Brewster JA, Garland EM, Biaggioni I, Black BK, Ling JF, Shibao CA,etal. Diurnal variability in orthostatic tachycardia: implications for the postural tachycardia syndrome. Clin Sci (Lond). 2012;122: 25–31.
Raj SR, Biaggioni I, Yamhure PC, Black BK, Paranjape SY, Byrne DW, Robertson D. Renin-aldosterone paradox and perturbed blood volume regulation underlying postural tachycardia syndrome. Circulation. 2005 Apr 5;111(13):1574-82. doi: 10.1161/01.CIR.0000160356.97313.5D. Epub 2005 Mar 21. PMID: 15781744.
Greg Samsa, David Edelman, Margaret L. Rothman, G. Rhys Williams, Joseph Lipscomb and David Matchar, Determining Clinically Important Differences in Health Status Measures A General Approach with Illustration to the Health Utilities Index Mark II 10.2165/00019053-199915020-00003
Vitez C, Blum CL. Postural orthostatic tachycardia syndrome (POTS) in a patient as a consequence of a concussion along with occipital headaches, dizziness and nausea: A case report. Asia-Pac Chiropr J. 2022;3.1 URL apcj.net/papers-issue-3-1/#VitezBlumTachycardia
Brett S Jarosz, Fellow Sports & Exercise Chiropractor (AICE 2019); Private Practice of Chiropractic, South Yarra, Victoria.Goodkin MB, Bellew LJ. Osteopathic manipulative treatment for postural orthostatic tachycardia syndrome. J Am Osteopath Assoc. 2014 Nov;114(11):874-7. doi: 10.7556/jaoa.2014.173. PMID: 25352409.
Budgell BS . Modulation of visceral function by somatic stimulation. In:KingHH,JanigW,PattersonMM, eds.The Science and Clinical Application of Manual Therapy. Churchill Livingstone/Elsevier; 2011:77-80.
Wagoner AL, Olson JD, Westwood BM et al (2019) Children with orthostatic intolerance exhibit elevated markers of inflammation in the dorsal medulla. Am J Physiol Heart Circ Physiol 317:H323–H329
Svetlana Blitshteyn, Is postural orthostatic tachycardia syndrome (POTS) a central nervous system disorder? Journal of Neurology https://doi.org/10.1007/s00415-021-10502-z
Freeman R, Weiling W, Axelrod F et al (2011) Consensus statement on the definition of orthostatic hypotension, neurally mediated syncope and the postural tachycardia syndrome. Clin Auton Res 21:69–72
De Wandele I, Rombaut L, Leybaert L, Van de Borne P, De Backer T, Malfait F, De Paepe A, Calders P. Dysautonomia and its underlying mechanisms in the hypermobility type of Ehlers-Danlos syndrome. Semin Arthritis Rheum. 2014 Aug;44(1):93-100. doi: 10.1016/j.semarthrit.2013.12.006. Epub 2013 Dec 30. PMID: 24507822.
Low PA, Sandroni P, Joyner M, Shen WK. Postural tachycardia syndrome (POTS). J Cardiovasc Electrophysiol. 2009 Mar;20(3):352-8. doi: 10.1111/j.1540-8167.2008.01407.x. Epub 2009 Jan 16. PMID: 19207771; PMCID: PMC3904426.
Wang E, Ganti T, Vaou E, Hohler A. The relationship between mast cell activation syndrome, postural tachycardia syndrome, and Ehlers-Danlos syndrome. Allergy Asthma Proc. 2021 May 1;42(3):243-246. doi: 10.2500/aap.2021.42.210022. PMID: 33980338.
Lambert E, Eikelis N, Esler M, Dawood T, Schlaich M, Bayles R, Socratous F, Agrotis A, Jennings G, Lambert G, Vaddadi G. Altered sympathetic nervous reactivity and norepinephrine transporter expression in patients with postural tachycardia syndrome. Circ Arrhythm Electrophysiol. 2008 Jun 1;1(2):103-9. doi: 10.1161/CIRCEP.107.750471. Epub 2008 Apr 30. PMID: 19808400.
Fedorowski A. Postural orthostatic tachycardia syndrome: clinical presentation, aetiology and management. J Intern Med. 2019 Apr;285(4):352-366. doi: 10.1111/joim.12852. Epub 2018 Nov 23. PMID: 30372565.
Mar PL, Raj SR. Postural Orthostatic Tachycardia Syndrome: Mechanisms and New Therapies. Annu Rev Med. 2020 Jan 27;71:235-248. doi: 10.1146/annurev-med-041818-011630. Epub 2019 Aug 14. PMID: 31412221.
Li H, Yu X, Liles C, Khan M, Vanderlinde-Wood M, Galloway A, Zillner C, Benbrook A, Reim S, Collier D, Hill MA, Raj SR, Okamoto LE, Cunningham MW, Aston CE, Kem DC. Autoimmune basis for postural tachycardia syndrome. J Am Heart Assoc. 2014 Feb 26;3(1):e000755. doi: 10.1161/JAHA.113.000755. PMID: 24572257; PMCID: PMC3959717.
Ware JE, Jr, Snow KK, Kosinski M, Gandek B. SF-36 Health Survey Manual and Interpretation Guide. Boston, MA: The Health Institute, New England Medical Centre; 1993
Schondorf, R., Low, P.A., 1993. Idiopathic postural orthostatic tachycardia syndrome: an attenuated form of acute pandysautonomia? Neurology 43, 132–137. https://doi. org/10.1212/wnl.43.1_part_1.132.
Jackson, G.L., Powers, B.J., Chatterjee, R., Bettger, J.P., Kemper, A.R., Hasselblad, V., Dolor, R.J., Irvine, R.J., Heidenfelder, B.L., Kendrick, A.S., Gray, R., Williams, J.W., 2013. The patient centered medical home. A systematic review. Ann. Intern. Med. 158, 169–178. https://doi.org/10.7326/0003-4819-158-3-201302050-00579.
Williams, J.W., Jackson, G.L., Powers, B.J., Chatterjee, R., Bettger, J.P., Kemper, A.R., Hasselblad, V., Dolor, R.J., Irvine, R.J., Heidenfelder, B.L., Kendrick, A.S., Gray, R., 2012. Closing the quality gap: revisiting the state of the science (vol. 2: the patient- centered medical home). Evid. Rep. Technol. Assess. 1–210 (Full. Rep).
Brazier JE, Harper R, Jones NM, O'Cathain A, Thomas KJ, Usherwood T, Westlake L. Validating the SF-36 health survey questionnaire: new outcome measure for primary care. BMJ. 1992 Jul 18;305(6846):160-4. doi: 10.1136/bmj.305.6846.160. PMID: 1285753; PMCID: PMC1883187.Raj SR. Postural tachycardia syndrome (POTS). Circulation. 2013;127:2336–42.
DaCosta JM. On irritable heart: a clinical study of a form of functional cardiac disorder and its consequences. Am J Med Sci. 1871;61:17–52.
Adena Zadourian · Taylor A. Doherty · Iwona Swiatkiewicz, · Pam R. Taub,Postural Orthostatic Tachycardia Syndrome: Prevalence, Pathophysiology, and Management https://doi.org/10.1007/s40265-018-0931-5
Michael T. Burcon, B.Ph., D.C.Upper Cervical Specific Pattern Analysis Utilizing Paraspinal Thermography, Leg Length Inequality and Heart Rate Variability in Two Patients with Tachycardia Journal of Upper Cervical Chiropractic Research ~ March 17, 2016 ~ Pages 6-12
Jeremy Wilson, DC Resolution of Chronic Postural Orthostatic Tachycardia Syndrome (POTS) & Dysautonomia Following Chiropractic Care: A Case Study & Review of Literature, Annals of Vertebral Subluxation Research ~ July 24, 2022 ~ Volume 2022 ~ Pages 91-96